Understanding the Blueprint
A well-designed bedroom house plan lays the foundation for a comfortable and functional living space tailored to your needs and preferences. From spacious master suites to cozy guest rooms, each bedroom plays a vital role in shaping the overall layout and flow of the home.
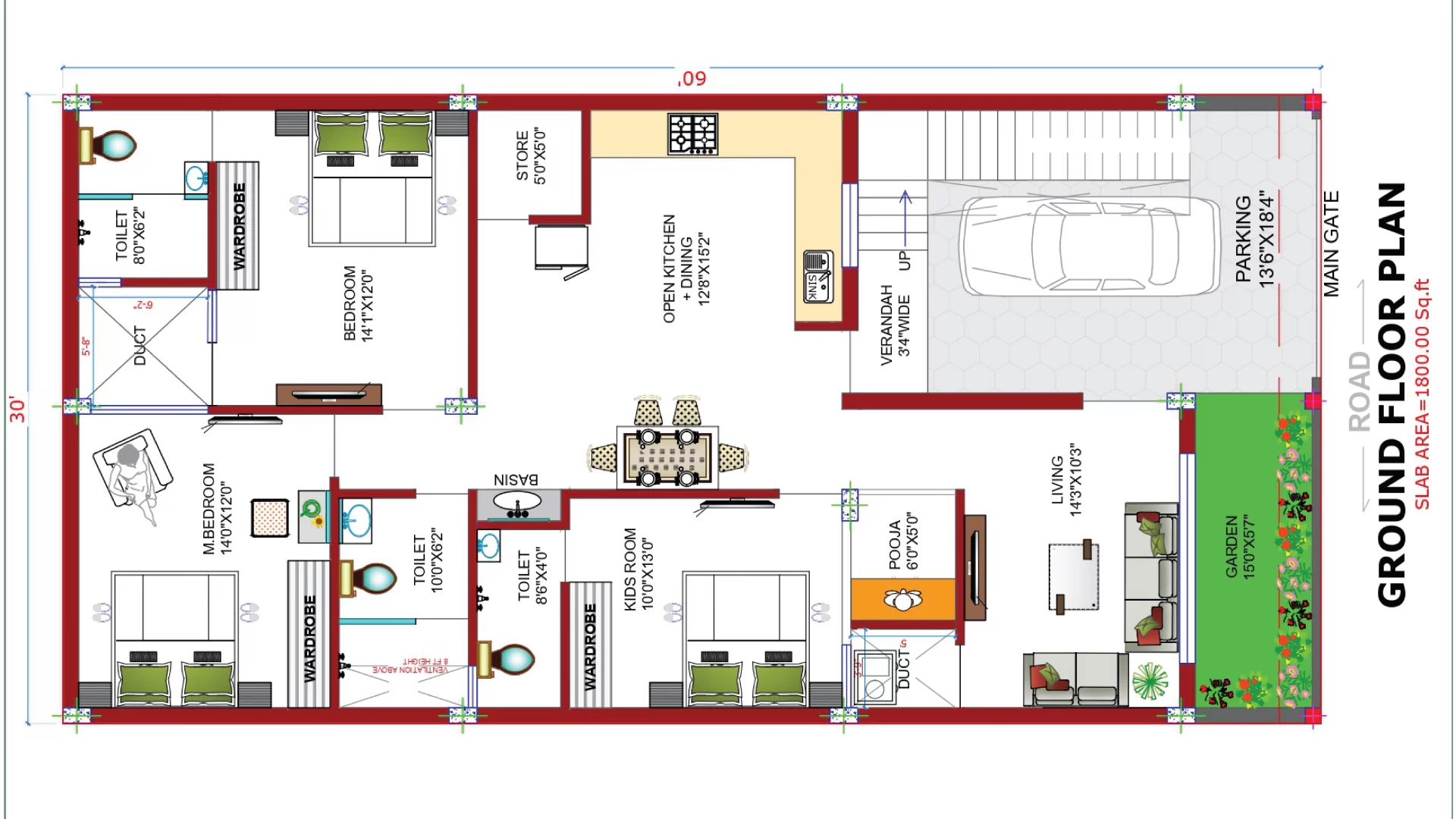
Mastering the Art of Bedroom Design
1. Master Suite:
The master bedroom serves as a sanctuary for relaxation and rejuvenation, featuring ample space for a king or queen-sized bed, bedside tables, and additional seating areas. Ensuite bathrooms with luxurious amenities such as soaking tubs, walk-in showers, and double vanities add a touch of elegance and convenience to the master suite.
2. Guest Bedrooms:
Guest bedrooms provide comfortable accommodations for overnight visitors, family members, or friends. These rooms may vary in size and configuration depending on the needs of the homeowner, offering flexibility to accommodate different sleeping arrangements and preferences.








